There are doubts about the effectiveness of masks, lockdowns, health passes, social distancing, vaccines, modes of transmission, incubation period, infectivity of asymptomatic persons, relevance of PCR tests, but the foundation, the virus, is indisputable.
It is settled, everyone knows it, there are thousands of studies, SARS-CoV-2 is the virus responsible for the symptom grid called COVID-19.
There are moreover three studies that claim to demonstrate that this virus is contagious and responsible for a disease, in animals of course, ethics dictates, but if proof is needed for those who doubt (who would doubt the obvious?), it is in these articles that we will find it.
Mice
The first is a Chinese study published on May 07, 2020 in the journal Nature, “The pathogenicity of SARS-CoV-2 in hACE2 transgenic mice“, it claims to “have confirmed the pathogenicity of SARS-CoV-2 in hACE2 mice”.
The hACE2 mice are genetically engineered to express human ACE2 (hACE2), an angiotensin 2 converting enzyme present in virtually every organ in humans and identified as the functional receptor for SARS.
The experiment is simple, there are three groups of mice:
The first group consists of transgenic mice (hACE2) which are inoculated nasally after anesthesia with Avertin with the SARS-CoV-2 isolate (an isolate in virology is not an isolate as in bacteriology, the isolate is here a cell culture on Vero monkey kidney cells containing L-glutamine, glucose, sodium pyruvate, sodium bicarbonate, fetal bovine serum, penicillin and streptomycin);
The second group consists of normal wild-type mice that are inoculated with the isolate in the same manner;
The third group consists of transgenic mice that are nasally inoculated with a saline solution (phosphate-buffered saline).
Slight bristling and weight loss was observed only in inoculated hACE2 mice, reaching 8% 5 days after infection and then rapidly returning to normal, no other clinical signs were detected.
If bristling hair and weight loss do not necessarily correspond to the symptoms observed in humans, these are mice and it is possible that they react differently.
However, this experiment has some shortcomings that may cast doubt on the relevance of the results obtained.
First, in the control experiment, the scientists claim to use an isolate, but the term is not appropriate since the solution is more comparable to a soup of genetic material from three different species and toxic products, this solution does not contain only the virus. It would be very simple to reproduce the same cell culture without adding the SARS-CoV-2 isolate and inoculate the control group, yet only a saline solution is used.
Would the same results have been obtained if the control mice had been treated in the same way?
Perhaps the scientists did not think it was worthwhile to do this experiment, because they also inoculated wild-type mice in the same way as the transgenic mice, and they did not show any symptoms (weight loss and bristly fur). But since the control experiment with the transgenic mice is questionable, it is not established that the cell culture products alone do not cause a different reaction between wild-type and hACE2 mice.
Any genetic modification leads to unpredictable reactions, e.g. transgenic mice overexpressing ACE2 in the heart are known to show sudden death due to cardiac arrhythmias. The possible explanation for this unfavorable phenotype may be related to an undefined role of CEA2 during development. It is likely that chronic overexpression of ACE2 and/or Ang-(1-7) may induce developmental defects in transgenic mice.
The results of this study using genetically modified mice are therefore difficult to interpret, it is possible that the methods alone used in this experiment, apart from the virus provided by another team of researchers, lead to the same results.
This study also caused a scandal when it was first published, because manipulations in the histopathological photographs were detected by Elisabeth Bik, a consultant in microbiome and scientific integrity.
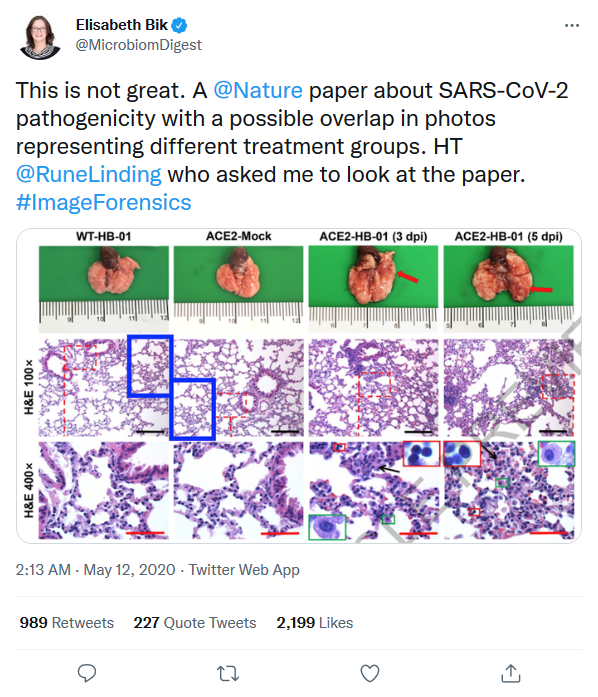
Figure 3 also raises questions,
“we demonstrated the colocalization of SARS-CoV-2 S protein (Fig. 3f) and the human ACE2 receptor (Fig. 3g) in alveolar epithelial cells of HB-01-infected hACE2 mice using immunofluorescence, at 3 dpi (Fig. 3h). This phenomenon was not found in mock-treated [with saline] hACE2 mice (Fig. 3a–d) or HB-01-infected wild-type mice (data not shown), which indicates that SARS-CoV-2—as with SARS-CoV—uses the human ACE2 as a receptor for entry.”

It would have been more relevant to compare the results of the inoculated wild-type mice with those of the transgenic mice to demonstrate this point, it is quite possible that the S protein is detected simply because it has been inoculated, independently of the hACE2 receptor, and in this case a picture of an inoculated wild-type mouse in which the S protein would not be detected would give more credibility to the study.
This study therefore hardly provides irrefutable evidence of the link between the genetic material used and the weight loss and bristly fur of the transgenic mice, but other studies of this type may be able to answer the different points raised.
Hamsters
The second paper is a Chinese study published on December 3, 2020, “Simulation of the Clinical and Pathological Manifestations of Coronavirus Disease 2019 (COVID-19) in a Golden Syrian Hamster Model: Implications for Disease Pathogenesis and Transmissibility“.
This breed of hamster was chosen because of the similarity between human and animal ACE2.
The experiment uses similar methods to the first one on mice, a group of 11 is inoculated intranasally with the SARS-CoV-2 “isolate”, and a group of 3 with a saline solution (PBS). Only the anesthetics change, here ketamine and xylazine.
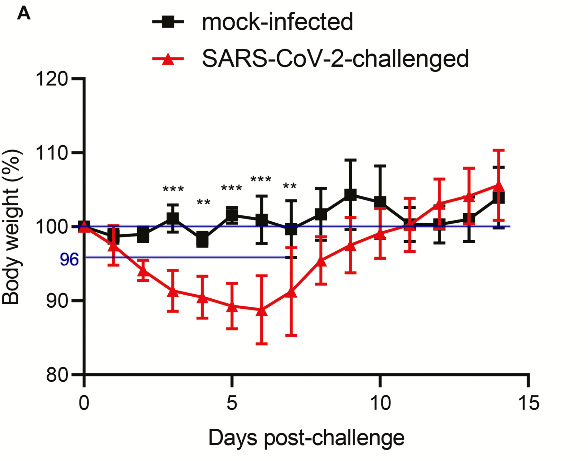
“SARS-CoV-2–challenged [cell culture/isolate] but not mock-infected animals [saline] exhibited progressive mean body weight loss of up to approximately 11% from 1 dpi to 6 dpi, then gradually regained their weight by 14 dpi (Figure 2A). They developed lethargy, ruffled fur, hunched back posture, and rapid breathing since 2 dpi and started to recover at 7 dpi. None of the SARS-CoV-2–infected and mock-infected animals died.”
This study presents the same flaws as the first one concerning the control experiment, there is also an important difference in the number of hamsters used (11 versus 3) inducing a variation of the weight parameters between the average of the two groups and an absence of control group for the histopathological images of the respiratory tract (available for the spleen and the intestine, which is of little interest)
In the second part of the study, hamsters inoculated (n=3) with the isolate were placed in the same cage as healthy non-inoculated hamsters (n=3) to study infectivity.
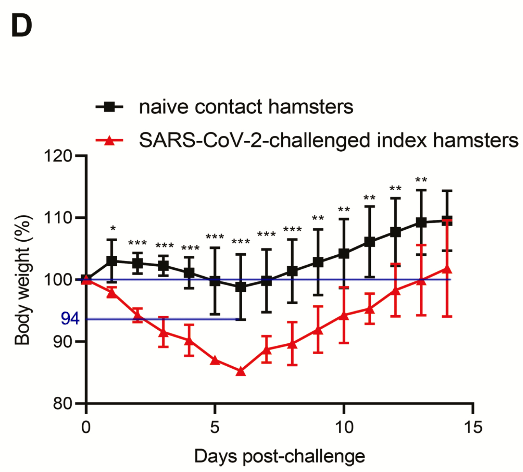
The inoculated group shows a progressive average body weight loss of up to about 15% from 1 to 6 days, the naive contact group does not show a greater average weight loss than the saline inoculated group, in fact they gain significantly more weight after 14 days.
It is noted, “Interestingly, the index animals exhibited significantly more weight loss (P < .05) than the contact animals between 2 and 8 dpi.”
“The minimal amount of weight loss in the naive contact animals could be related to a lower virus inoculum when compared with the high single intranasal virus dose received by the challenged index animals.”
The scientists do not mention at any time the clinical signs of the naive contact hamsters, perhaps they did not observe any.
They conclude that the experiment worked, because both groups of hamsters tested positive by PCR test, but there is no evidence that the contact hamsters became ill. It is possible that some of the genetic material inoculated into the index hamsters spread to the contact hamsters without inducing any symptoms, and that the challenged index hamsters showed symptoms because of the methods and products contained in the isolate.
It is also stated that “The histopathological changes and viral N protein expression in the contact animals’ nasal turbinate, trachea, lung, and extrapulmonary tissues were similar to those of the challenged animals (Supplementary Figure 5).
Supplementary Figure 5 in question:
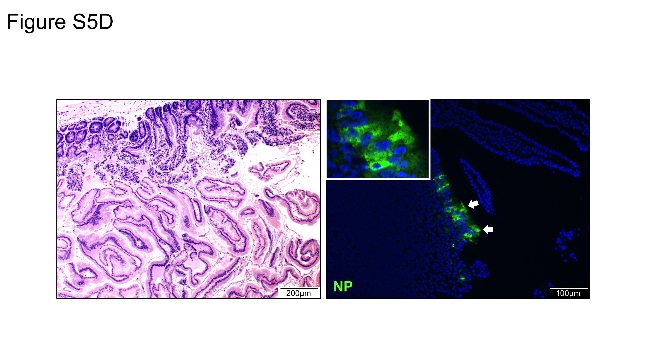
There is no precision on the part of the respiratory tract represented, and the N gene seems to be present in trace amounts compared to the other images.
This second study therefore hardly shows the link between SARS-CoV-2 and a disease in hamsters, the experience of transmissions between inoculated hamsters and contact hamsters even tends to demonstrate a lack of infectivity between contacts (in hamsters).
Macaques
The third study, published on May 12, 2020 by a team in the United States, “Respiratory disease in rhesus macaques inoculated with SARS-CoV-2” is the perfect model to study the contagiousness of SARS-CoV-2 in a non-human model, as the rhesus macaque ACE2 is 100% identical to human ACE2 at the interface region.
8 macaques (4 males and 4 females) are inoculated with a cell culture of similar composition to the isolates of the first and second study via a combination of intranasal (0.5 ml per nostril), intratracheal (4 ml), oral (1 ml) and ocular (0.25 ml per eye) administration.
“We inoculated eight adult rhesus macaques with the SARS-CoV-2 isolate nCoV-WA1-202014. On day 1 post-inoculation (dpi), all macaques showed changes in their respiratory pattern and piloerection [bristling of the hairs], as reflected in their clinical scores (Fig. 1a). Other signs of disease we observed included reduced appetite, a hunched posture, pale appearance and dehydration (Extended Data Table 1). Coughing was occasionally heard in the room where macaques were housed, but could not be pinpointed to individual macaques. Signs of disease persisted for more than a week, with all macaques being completely recovered between 9 and 17 dpi (Fig. 1a, Extended Data Table 1). We observed weight loss in all macaques (Fig. 1b); body temperatures spiked at 1 dpi but returned to normal levels thereafter (Fig. 1c). Under anaesthesia, the macaques did not show increased respiration; however, all macaques showed irregular respiration patterns (Fig. 1d).”
We first note that no control experiment is performed, “Blinding was not used in this study as all macaques were subjected to the same treatment.”, a questionable logic, the treatment to which the macaques are subjected being potentially traumatic (the macaques are not anaesthetized and surely restrained for the inoculation) and the cell culture used containing many other substances than the SARS-CoV-2 virus, it could potentially induce a toxic or allergic reaction.
The scientists themselves point out that the breathing patterns of the macaques return to normal after anesthesia (for euthanasia), indicating that the change in breathing, as well as the other symptoms, could also be related to the stress induced by the physical methods used for the inoculation.

The macaques are also tested by PCR:
“Virus shedding was highest from the nose (Fig. 2a); virus could be isolated from swabs collected at 1 and 3 dpi, but not thereafter. Viral loads were high in throat swabs immediately after inoculation, but were less consistent than nose swabs thereafter; in one macaque, throat swabs were positive at 1 dpi and at 10 dpi—but not in between (Fig. 2a).”
These data suggest that inoculated cell culture results in a positive PCR test if performed on the inoculated tissue. Putting genetic material into macaques and finding that genetic material in them thus proves that genetic material has been inoculated into macaques. The “viral load” does not seem to increase as one would expect if the virus was replicating in the cells, it seems to be excreted as an exogenous particle that cannot be assimilated by the organism (of the macaque).
Histopathological and lung images are difficult to interpret in the absence of a control group subjected to the same treatment (but without virus). Lung inhalation of the cell culture could also cause the observed “changes”.
We have therefore reviewed here the three major scientific papers used to demonstrate the pathogenicity of SARS-CoV-2 and its causal link to the Covid-19 disease.
If they have some methodological flaws, it still does not prove that SARS-CoV-2 is not contagious and responsible for COVID-19 in humans.
Why is this so?
Because we have no other model to explain disease, biology and life. A so-called infectious disease must be caused by a unique material agent, and the elimination of this element of unknown origin (a bat, a pangolin, the neighbor, who themselves “caught the virus” following its formation through random chemical reactions of nature and the universe) can only be achieved by a seek & destroy approach. The virus is a unique, random, purposeless entity that the body’s cells replicate until they burst, that’s just the way it is.
If nutrition, deficiencies, psychological state, microbiome, toxins and pollutants have a role on health, it is above all by affecting the immune system which allows us to fight against pathogenic viruses and bacteria. Humans have evolved over thousands of years by adapting to viruses, to the point that 8% of their DNA is composed of them, they are an integral part of our genetic code.
It is therefore impossible that SARS-CoV-2 is not contagious, it is impossible that SARS-CoV-2 is not responsible for the symptom grid called COVID-19, because it is impossible that we think in a different way.
Antonin Pimousse


